|
However, chemical processes can be either reversible or irreversible. Entropy can be mathematically expressed as the division between the change in heat and temperature. Entropy is symbolized by ‘S’, and as it is a state function, it is always written as a capital letter. It only depends on the beginning and the ending points of a reaction. Therefore, if a reaction has a higher number of molecules as products than its reactants, it can be concluded that the reaction is moving towards a higher level of disorder, and this is a favourable condition in chemistry.Įntropy is identified as a state function in chemistry as the change in entropy doesn’t depend on the chemical path travelled when moving from the chemical reactants to the products. The randomness or the disorder of a reaction increases as the number of molecules increases. What this means is that chemical reactions will tend to drive themselves in the direction of more disorder. According to the second law of thermodynamics, it is believed that the entropy of an isolated system will always increase. What is EntropyĪs mentioned above, entropy measures the randomness or the extent of disorder in a chemical process. 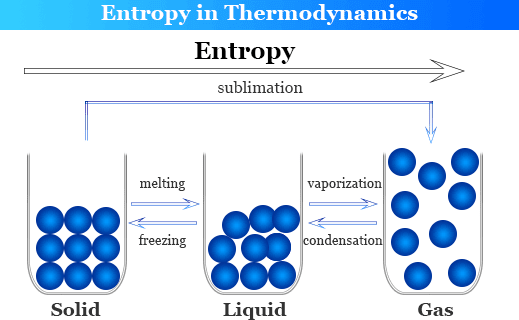
The main difference between entropy and enthalpy is, entropy is used as a measurement of the disorder or the randomness of a chemical process while enthalpy is used as a measure of the heat change of a chemical reaction or the change in internal energy of a reaction under constant pressure. And also they are both related to the heat changes of a reaction. Both Entropy and Enthalpy relate to functions of measurements in chemical thermodynamics.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
